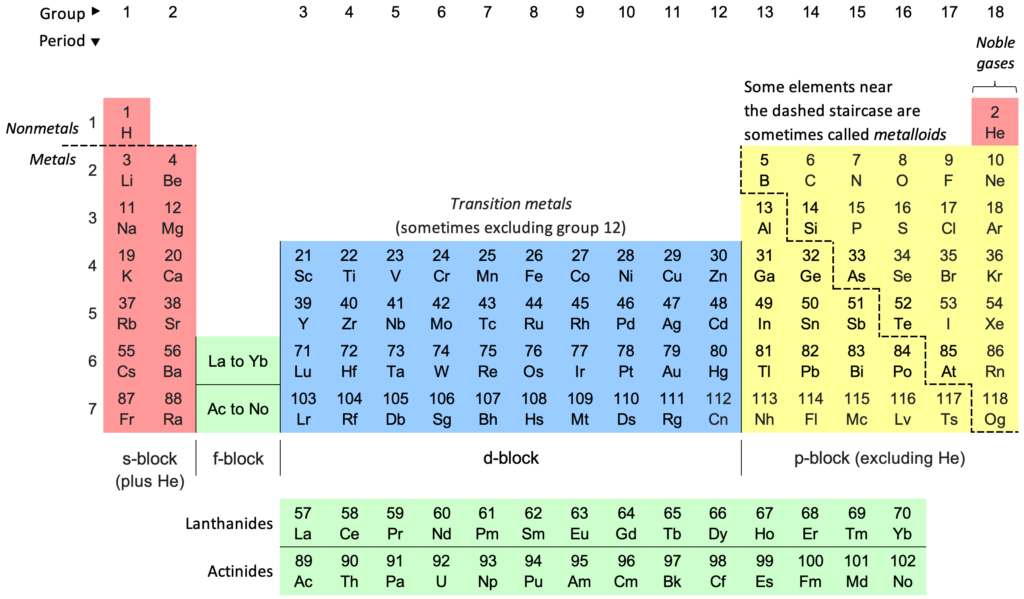
Solution:As we move from left to right across a period, Chemical reactivity - First decreases and then increases, Nuclear charge - Increases, Atomic number - increases, Valence electronsincreases, Atomic radii - decreases. As we move down a group, Chemical reactivity- Increases, Atomic numberincreases, Valence electrons- no change, Atomic radii - Decreases.