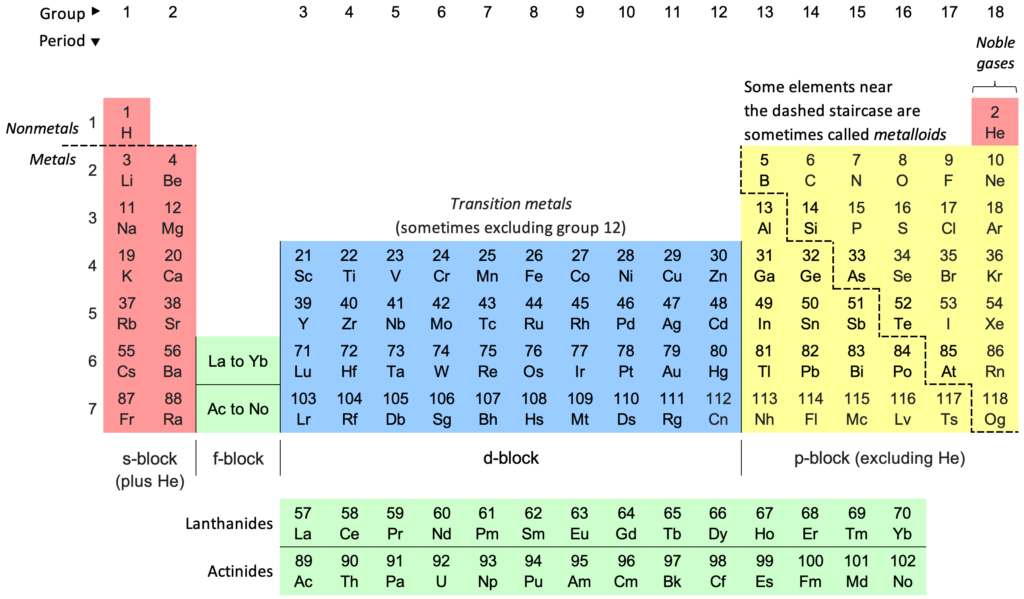
Solution:Elements (Atomic number, Valency) - Aluminium (Al) (13, 3) < Silicon (Si) (14, 4) < Phosphorus (P) (15, 3 ) < Sulfur (S) (16, 2) < Chlorine (Cl) (17, 1). Lithium (Li) (3, 1), Beryllium (Be) (4, 2), Boron (B) (5, 3), Carbon (C) (6, 4), Nitrogen (N) (7,3), Sodium (Na) (11, 1), Magnesium (Mg) (12, 2), Argon (Ar) (18, 0), Fluorine (F) (9, 1), Neon (Ne) (10, 0).
The electrons present in the outermost shell of an atom are called valence electrons. Valency - number of electrons required to complete octet. There are four types of orbital present in atoms - s, p, d, f.