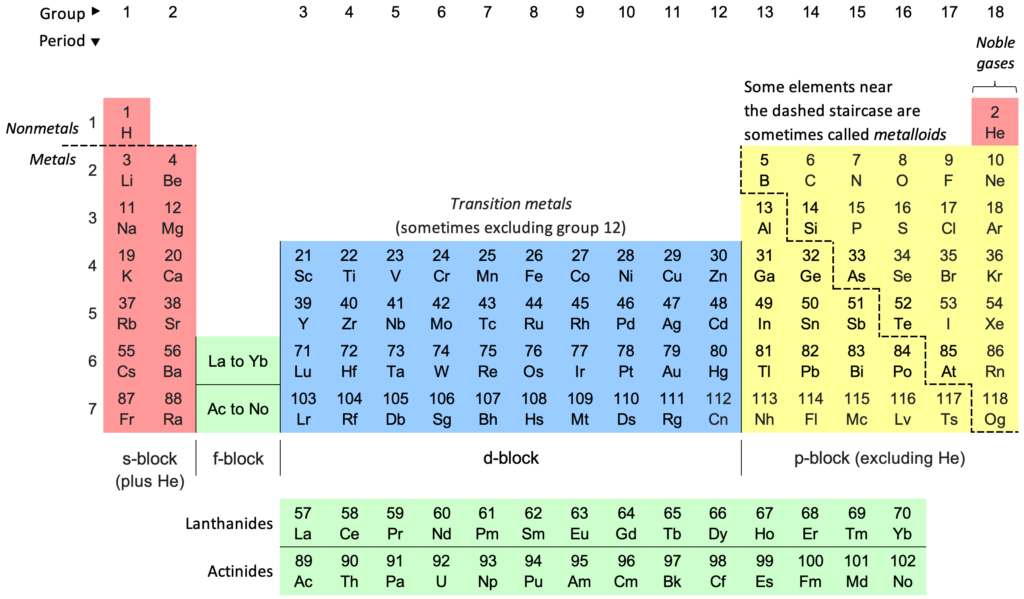
Solution:Dobereiner’s triad - Groups of three elements (triads) with similar physical and chemical properties where the arithmetic mean of the atomic masses of the first and third element in a triad would be approximately equal to the atomic mass of the second element.
Examples - {lithium (Li), Sodium (Na) and Potassium (K)}, {Calcium (Ca), Barium (Ba) and Strontium (Sr)}, {Chlorine (Cl), Bromine (Br) and Iodine (I)}, {Sulphur (S), Selenium (Se), and Tellurium (Te)}, {Iron (Fe), Cobalt (Co) and Nickel (Ni)}.