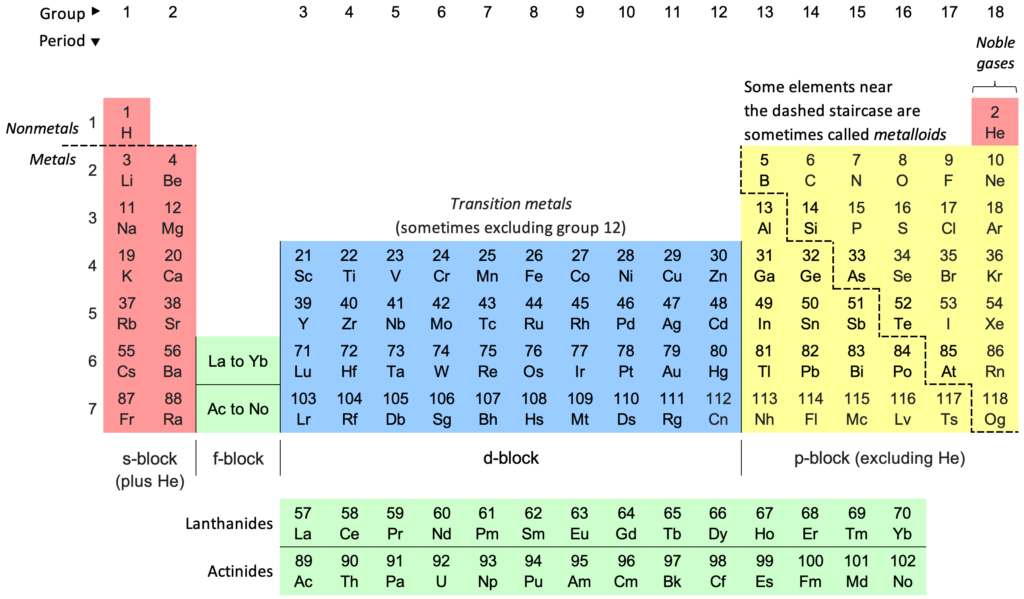
Solution:In a period from left to right, nuclear charge increases. Therefore, F has the highest nuclear charge among all, and on moving down the group, the effective nuclear charge decreases due to addition of electrons in the shells moving away from the nucleus. In the Modern periodic table, Na lies below Li in same group (alkali metals) hence, it has the least nuclear charge among the given options.