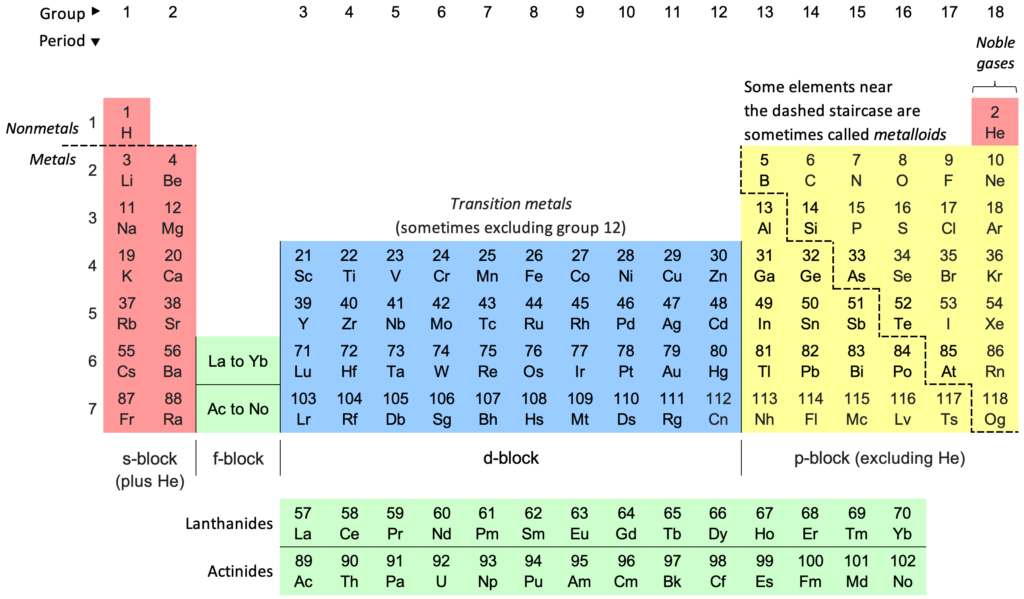
Solution:The elements having atomic numbers: 1 - Hydrogen (H), 2 - Helium (He), 3 - Lithium (Li), 4 - Beryllium (Be), 5 - Boron (B), 6 - Carbon (C), 7 - Nitrogen (N), 8 - Oxygen (O), 9 - Fluorine (F), 10 - Neon (Ne), 11 - Sodium (Na), 12 - Magnesium (Mg), 13 - Aluminum (Al), 14 - Silicon (Si), 15 - Phosphorus (P), 16 - Sulfur (S), 17 - Chlorine (Cl), 18 - Argon (Ar), 19 - Potassium (K), 20 - Calcium (Ca).